.svg/500px-Periodic_table_(polyatomic).svg.png)
Write the valence electron configuration of each element by first indicating the filled inner shells using the symbol for the nearest preceding noble gas and then listing the principal quantum number of its valence shell, its valence orbitals, and the number of valence electrons in each orbital as superscripts.Ī The group 2 elements are in the s block of the periodic table, and as group 2 elements, they all have two valence electrons.  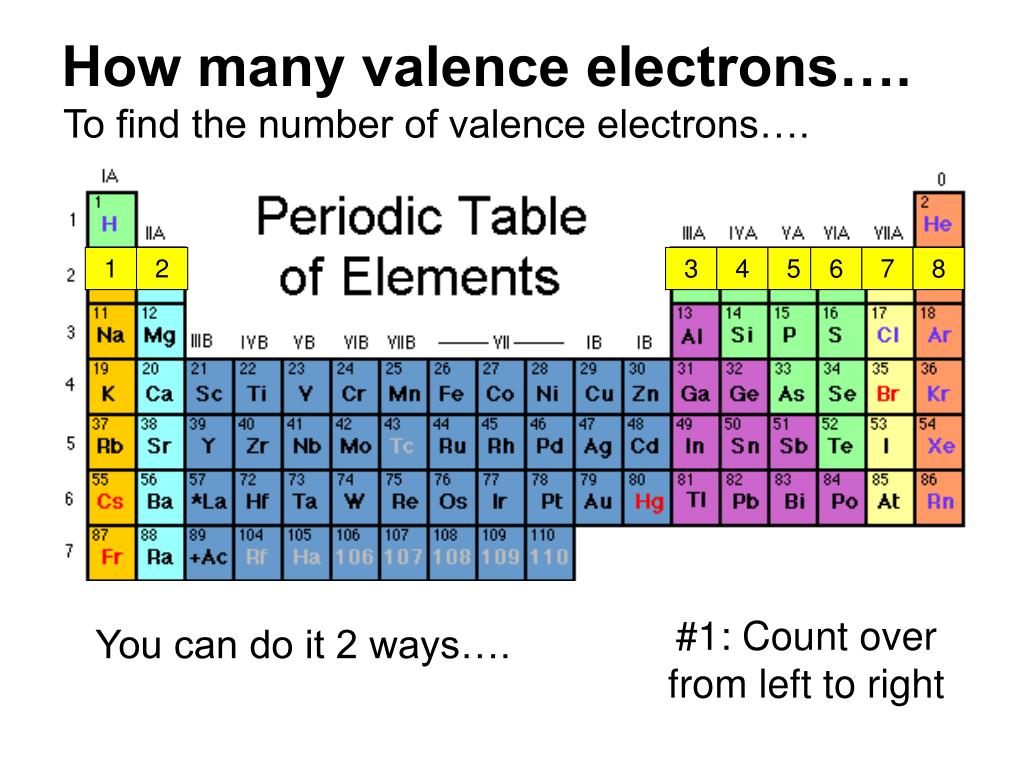
Locate the nearest noble gas preceding each element and identify the principal quantum number of the valence shell of each element. For example, the elements in the first column all have a single valence electron, an electron that can be donated in a chemical reaction with another atom. These electrons are known as valence electrons. Identify the block in the periodic table to which the group 2 elements belong. In the periodic table of the elements, elements in a single column have the same number of electrons that can participate in a chemical reaction.Use the periodic table to predict the valence electron configuration of all the elements of group 2 (beryllium, magnesium, calcium, strontium, barium, and radium).Īsked for: valence electron configurations For elements after No, the electron configurations are tentative. The electron configurations of the elements indicated in blue are also anomalous, but the reasons for the observed configurations are more complex. However, there are also other patterns in chemical properties on the periodic table. This similarity occurs because the members of a group have the same number and distribution of electrons in their valence shells. How to Find the Number of Valence Electrons for Transition Metals. The elements in groups (vertical columns) of the periodic table exhibit similar chemical behavior. The electron configurations of elements indicated in red are exceptions due to the added stability associated with half-filled and filled subshells. How to Determine Number of Valence Electrons in Element, Ion, and Compound (Count Valence Electrons) Conquer Chemistry. \): Electron Configurations of the Elements.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |



 RSS Feed
RSS Feed
